 Schematics of oxidized catechol conjugation with unpaired thiol in protein
Schematics of oxidized catechol conjugation with unpaired thiol in proteinAbstract
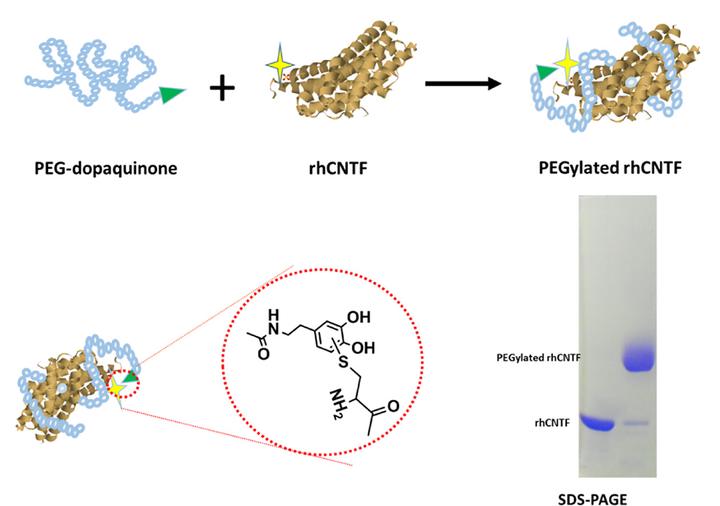
Site-specific PEGylation of clinically used protein via free cysteine residue has been widely applied for a homogeneous product. Herein, we described a new thiol-specific PEGylation strategy based on catechol-derived reactive quinone species. Catechol-derived polyethylene glycol (PEG) was synthesized by coupling linear PEG N-hydroxysuccinimide to dopamine and then oxidized to quinone. PEG-dopaquinone (PEG-DAQ) mostly modifying the free cysteines of two model proteins of truncated flagellin (CBLB502) and recombined human ciliary neurotrophic factor (rhCNTF) evidenced its thiol-specificity. The yield of PEG-DAQ-rhCNTF under neutral pH value was 87.5%, compared with PEG-maleimide of 92.3% and PEG-vinylsulfone of 17.6%, respectively. About 95% reactive capacity for PEG-DAQ was maintained after incubation in aqueous solvent for 96 h, indicating that the reagent was superiorly hydro-stable. Moreover, it was found that quenching the PEGylation reaction by excess cysteine was necessary to obtain more stable conjugate. The resulted PEG-DAQ-rhCNTF remained about 93.5% of the initial products after storage for 14 days. As a comparison, PEG-MAL-rhCNTF was remained to 71.2%. Our results demonstrated that PEG-dopaquinone had properties of high selectivity and reactivity with cysteine thiol, and superior stability for both the reagent itself and its conjugate, showing great promise for developing as an alternative reagent for thiol-selective modification.